Table of Contents
Week 1 | English Grammar
Day 4 |I am doing (present continuous)
Day 5 |Are you doing? (present continuous questions)
Day 6 |I do/work/like (present simple)(present continuous questions)
Week 2 | English Grammar
Day 1 |I don't... (present simple negative)
Day 2 |Do you...? (present simple questions)
Day 3 |I am doing (present continuous) I do (present simple)
Day 4 |I have... and I've got...
Day 6 |Worked/got/went etc (past simple)
Week 3 | English Grammar
Day 1 |I didn't... did you...? (past simple negative and questions)
Day 2 |I was doing (past continuous)
Day 3 |I was doing (past continuous) and I did (past simple)
Day 4 |I have done (present perfect 1)
Day 5 |I've just... I've already... I haven't...yet (present perfect 2)
Day 6 |Have you ever...? (present perfect 3)
Week 4| English Grammar
Day 1 |How long have you...? (present perfect 4)
Day 3 |I have done (present perfect) and I did (past)
Day 4 |Is done, was done (passive 1)
Day 5 |Is being done, has been done (passive 2)
Day 6 |Be/have/do in present and past tenses
Week 5| English Grammar
Day 1 |Regular and irregular verbs
Day 2 |What are you doing tomorrow?
Week 6| English Grammar
Day 2 |Must, mustn't, don't, need to
Day 6 |Do this! Don't do that! Let's do that
Week 7| English Grammar
Day 2 |There is... There are...
Day 3 |There was/were... There has/have been... There will be...
Day 6 |Have you? Are you? Don't you? etc
Week 8| English Grammar
Day 1 |Too/either/so am I/neither do I etc
Day 2 |Isn't/haven't/don't etc (negatives)
Day 3 |Do they? Is it? Have you?
Day 4 |Forming questions (who/what/why/where/when/which)
Day 5 |What...? Which...? How...?
Day 6 |How long does it take...?
Week 9| English Grammar
Day 1 |Do you know where...? I don't know what... etc
Day 2 |He/she said that... He/she told me that...
Day 3 |Work/working Go/going Do/doing
Day 4 |I want you to... I told you to...
Day 5 |I went to the shop to...
Day 6 |Go to... Go on... Go for... Go -ing... Get…
Week 10| English Grammar
Day 4 |I/me He/him They/them etc
Day 6 |Whose is this? It's mine/yours/hers etc
Week 11| English Grammar
Day 1 |Myself/yourself/themselves etc
Week 12| English Grammar
Day 2 |All/most/some/any/no/none etc
Week 13| English Grammar
Day 2 |If we go... if you see... etc
Day 3 |If I had... If we went... etc
Day 4 |A person who... A thing that/which (relative clauses 1)
The Periodic Table
The pH scale is a measure of how acidic or basic a solution is. It ranges from 0-14, with zero being the most acidic and 14 being the most alkaline. 7, being exactly in the middle, is what would be termed as neutral.
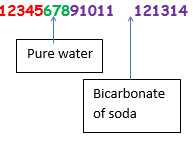
Any number less than seven is considered to be acidic – the lower the number, the more acidic the substance. 4 -6 would be considered ‘weakly acidic’.
Any number higher than seven is considered to be alkaline – the higher the number, the more alkaline the substance. 8-10 would be considered ‘weakly alkaline’
Substances which are very strongly acidic or alkaline tend to be reactive and can be even cause chemical burns, if not handled with care.
Measuring the pH
There are several different ways to measure the pH and find out whether a solution is acidic, alkaline or neutral. Some of these methods, such as Universal Indicator, will give an approximate numerical value on the pH scale, while others, such as Litmus paper, will only give a colour indication as to whether the solution is acidic, alkaline or neutral.
Universal Indicator
Universal indicator is a mix of several different indicators, which effectively allows it to show the whole range of different pHs.
It can be found in paper form, which is known as a pH test strip, or as a liquid solution which simply needs to be dropped onto a sample of the substance. After a couple minutes, there should be a colour change. The new colour should then be compared with the scale kit that is provided with the indicator and the pH can be estimated. Generally, the paper form is used when the solution is a dark colour already, while the liquid indicator is used on colourless samples.
Litmus Paper
Litmus paper can tell you if a solution is acidic or basic, but there is no ranging scale as there is in Universal Indicator. Instead, there will one of two colour changes to either red or blue and that is the result. If the Litmus paper is made wet, you can also measure the pH of gasses.
When Litmus paper is in neutral conditions, it will be purple.
When Litmus paper is in fairly strong acidic conditions (less than 4.5), it will be red.
When Litmus paper is in fairly strong alkaline conditions (more than 8.3), it will be blue.
KS3 Chemistry Questions – The pH Scale


1. What would 13 represent on the pH scale?
a. Acid
b. Alkali
c. Neutral
d. There’s no 13 on the pH scale
2. What number would represent a neutral substance?
a. 1
b. 4
c. 7
d. 10
e. 12
3. Which of these is NOT a form of Universal Indicator?
a. Paper strip
b. Gas
c. Liquid
4. Which of these is the most alkaline?
a. Bicarbonate of soda – pH 10
b. Water – pH 7
c. Sodium hydroxide – pH 12
d. Vinegar – pH 4
[bg_collapse view=”link” color=”#fafafa” expand_text=”Reveal Answer” collapse_text=”Hide Answer” inline_css=”background: #2ea3f2; padding: 9px; font-size: 14px; font-weight: 600;” ]
1.Alkali
2.7
3.Gas
4.Sodium hydroxide – pH 12
[/bg_collapse]

1. How does Litmus paper work?
2. Why is it important to know if something is strongly acidic or alkaline?
3. Lemon juice is a weak acid. Approximately where on the pH scale would it be found?
4. How is Universal Indicator different to Litmus paper?
[bg_collapse view=”link” color=”#fafafa” expand_text=”Reveal Answer” collapse_text=”Hide Answer” inline_css=”background: #2ea3f2; padding: 9px; font-size: 14px; font-weight: 600;” ]
1.A sample of the solution will be dropped onto the Litmus paper and the paper will either turn red or purple depending on it is acidic or basic. If the Litmus paper is made wet, it can measure the pH of gasses.
2.Strong acids and alkalis can be very reactive and cause chemical burns so it is important to know in order to handle with care.
The pH scale measures how acidic or alkaline a substance is.
3.Any answer given between 3 and 6 will be fine.
4.Universal indicator gives a numerical value of how acidic/alkaline something is. Litmus paper only tells you whether it is acidic or alkaline.
[/bg_collapse]

